 Women (16.2%) were also more likely to report adverse effects than men (9.3%) after the first dose of Pfizer, as was the case after AstraZeneca (39.3% women, 26.2% men). The phase 2–3 trial of the ChAdOx1 nCoV-19 vaccine reported systemic adverse effects in 88% of participants aged 18–55 years who received the first injection, whereas we found a lower rate of 33.7% after the first dose in the overall sample and 46.9% in individuals aged 18–55 years.’ ‘Similarly, rates of side effects following the ChAdOx1 nCoV-19 vaccine were lower than expected. However, in our community analysis, less than 30% of users complained of injection-site pain and less than 25% of fatigue and headache after the first dose,’ the authors wrote. ‘For example, in phase 3 clinical trials of the BNT162b2 vaccine, the most common events after the first dose were injection-site pain (71–83%), fatigue (34–47%), and headache (25–42%). 
However, researchers noted that side effects after both types of vaccine occurred at frequencies ‘lower than reported in phase 3 trials’. 
It found side effects are more common among women, people aged 55 years or younger, and after a second dose.įor example, 46.9% (AstraZeneca) and 20.7% (Pfizer) of people aged 55 years or younger reported at least one systemic effect after receiving their first dose, compared with 30.7% (AstraZeneca) and 10.6% (Pfizer) of those older than 55. The study also tested whether adverse effects varied across individuals’ characteristics, such as age and body mass index (BMI), gender, and health status. Fatigue and headaches were the most commonly reported systemic side effects, frequently reported within the first 24 hours after vaccination. The most frequently reported local side effects were tenderness and pain around the injection site, occurring most often the day after injection. Systemic side effects (diarrhoea, fatigue, headache, chills, nausea) affected fewer than one in four people, but were more common with AstraZeneca, with at least one symptom reported by 33.7% after the first dose, compared to 13.5% and 22% after the first and second respective dose of Pfizer. At least one symptom was reported by 71.9% of individuals after the first dose of Pfizer, 68.5% after the second dose, and by 58.7% after the first dose of AstraZeneca. They found local side effects (pain, swelling, redness, itching, swollen armpit glands) affected two in three people, and were more common in Pfizer recipients. Researchers had 627,383 people self-report their symptoms on the COVID Symptom Study app between 8 December and 10 March after receiving either their first or second dose of the Pfizer vaccine, or their first dose of AstraZeneca. 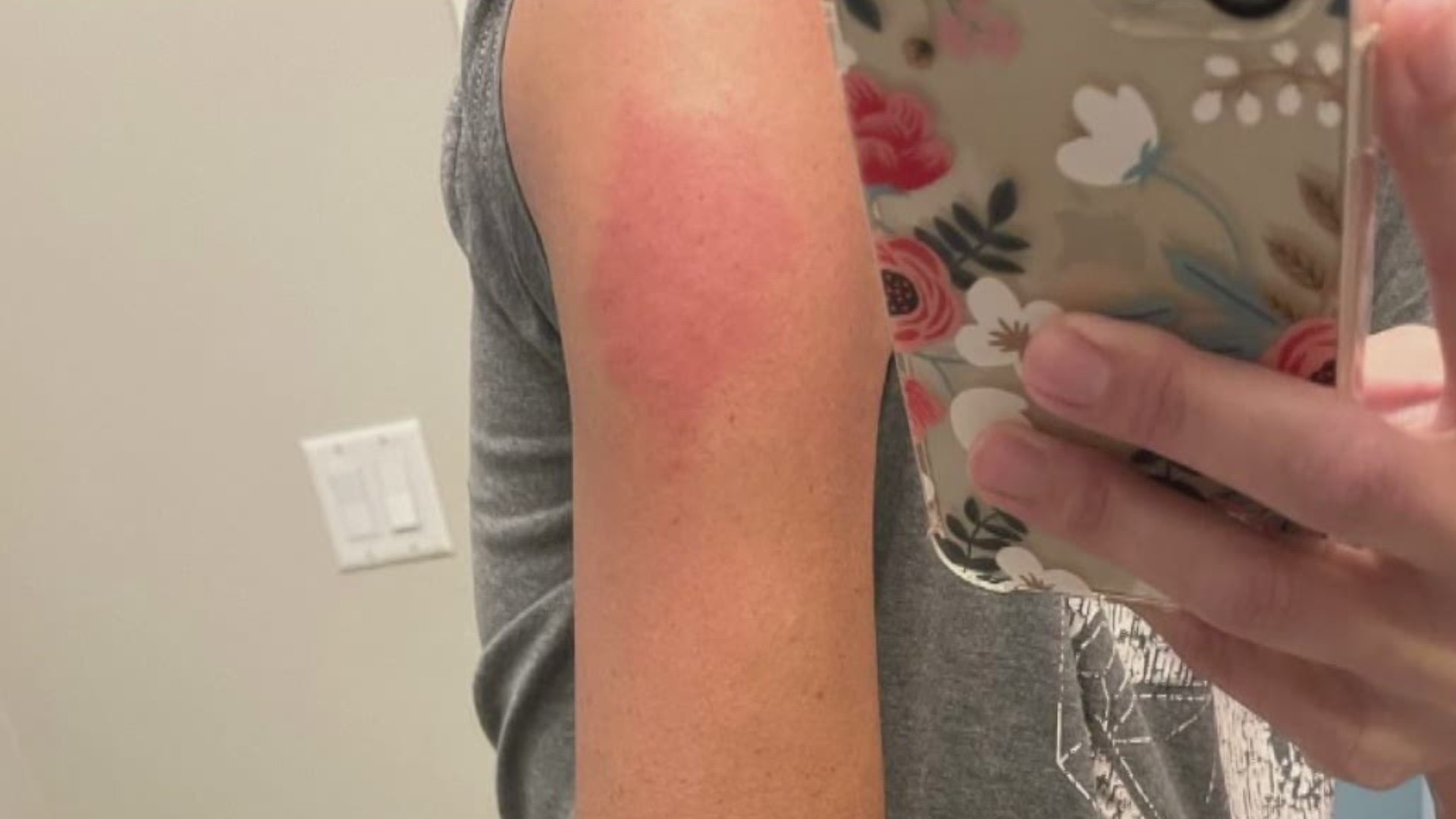
Now a new observational study from the UK, published in The Lancet, is giving insight into the typical side effects of the AstraZeneca and Pfizer vaccines, in addition to their effectiveness, in a community setting. Since then, more than one billion doses have been administered worldwide. It has been five months since the first vaccine against COVID-19 was administered to a 90-year-old woman in the UK. People under 55, women and those previously infected with COVID-19 are more likely to report side effects after receiving either vaccine, according to new research.Īmong recipients of the AstraZeneca and Pfizer vaccines, two in three report local side effects and one in four report systemic effects. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
